Nutrient Dynamics and Ecosystem Metabolism of Megacity Rivers: Influence of Elevated Nutrient Concentrations in Beijing’s Waterways
Abstract
:1. Introduction
2. Materials and Methods
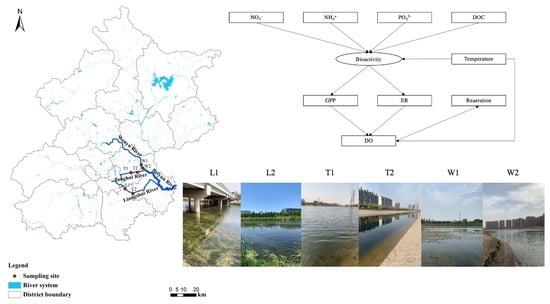
2.1. Study Area
2.2. Sample Collection
2.3. Concentration Measurement of NO3−, NH4+, PO43−, DOC
2.4. Flux Measurement of NO3−, NH4+, PO43−, DOC
2.5. Equilibrium Flux of NO3−, NH4+, PO43−, DOC
2.6. Sediment Analysis
2.7. Measurement of DO and Water Temperature
2.8. GPP, ER and Reaeration Estimates
2.9. Metabolism Data Standardization
2.10. Structural Equation Modelling
2.11. Data Analysis
3. Results
3.1. Nutrient Dynamics in Urban River Ecosystems
3.1.1. Water Quality of Study Rivers
3.1.2. Sediment Analysis
3.1.3. Nutrient Fluxes at the Sediment-Water Interface
3.2. Metabolic Patterns of Urban River Ecosystems
3.2.1. DO Profiles among Study Sites
3.2.2. GPP, ER and NEP Estimates
3.2.3. Reaeration and Activation Energy Estimates
3.2.4. SEM Pathway Analysis
4. Discussion
4.1. Nutrient Dynamics across the Sediment-Water Interface
4.1.1. Mechanisms Driving NO3− and NH4+ Concentrations and Fluxes
4.1.2. Mechanisms Driving Po43− Concentrations and Fluxes
4.1.3. Mechanisms Driving DOC Concentrations and Fluxes
4.1.4. Flux Predicted by Nutrient Concentrations and Water Quality
4.2. Metabolic Patterns of Urban River Ecosystems
4.2.1. DO Profiles across Study Sites
4.2.2. Factors Driving GPP and ER
4.2.3. Factors Driving Reaeration
4.3. Research Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Capps, K.A.; Bentsen, C.N.; Ramírez, A. Poverty, urbanization, and environmental degradation: Urban streams in the developing world. Freshw. Sci. 2016, 35, 429–435. [Google Scholar] [CrossRef]
- Jarvie, H.P.; Smith, D.R.; Norton, L.R.; Edwards, F.K.; Bowes, M.J.; King, S.M.; Scarlett, P.; Davies, S.; Dils, R.M.; Bachiller-Jareno, N. Phosphorus and nitrogen limitation and impairment of headwater streams relative to rivers in Great Britain: A national perspective on eutrophication. Sci. Total Environ. 2018, 621, 849–862. [Google Scholar] [CrossRef] [PubMed]
- Woodward, G.; Gessner, M.O.; Giller, P.S.; Gulis, V.; Hladyz, S.; Lecerf, A.; Malmqvist, B.; McKie, B.G.; Tiegs, S.D.; Cariss, H.; et al. Continental-scale effects of nutrient pollution on stream ecosystem functioning. Science 2012, 336, 1438–1440. [Google Scholar] [CrossRef] [PubMed]
- Booth, D.B.; Roy, A.H.; Smith, B.; Capps, K.A. Global perspectives on the urban stream syndrome. Freshw. Sci. 2016, 35, 412–420. [Google Scholar] [CrossRef]
- Liu, H.Q.; Kong, T.; Qiu, L.; Xu, R.; Li, F.; Kolton, M.; Lin, H.; Zhang, L.; Lin, L.; Chen, J.; et al. Solar-driven, self-sustainable electrolysis for treating eutrophic river water: Intensified nutrient removal and reshaped microbial communities. Sci. Total Environ. 2021, 764, 144293. [Google Scholar] [CrossRef]
- Garzon-Garcia, A.; Burton, J.; Franklin, H.M.; Moody, P.W.; De Hayr, R.W.; Burford, M.A. Indicators of phytoplankton response to particulate nutrient bioavailability in fresh and marine waters of the Great Barrier Reef. Sci. Total Environ. 2018, 636, 1416–1427. [Google Scholar] [CrossRef]
- Smith, A.J.; Duffy, B.; Onion, A.; Heitzman, D.; Lojpersberger, J.; Mosher, E.; Novak, M. Long-term trends in biological indicators and water quality in rivers and streams of New York State (1972–2012). River Res. Appl. 2018, 34, 442–450. [Google Scholar] [CrossRef]
- Lu, Q.; Xu, Y.; Xu, K.; Xia, W.; Liu, J.; Liu, Y. Experiments of water quality monitoring and sediment pollution release in Shamao River. Earth Environ. Sci. 2021, 668, 12039. [Google Scholar] [CrossRef]
- Smucker, N.J.; Detenbeck, N.E. Meta-Analysis of lost ecosystem attributes in urban streams and the effectiveness of out-of-channel management practices. Restor. Ecol. 2014, 22, 741–748. [Google Scholar] [CrossRef]
- Liu, X.C.; Beusen, A.H.; Van Beek, L.P.; Mogollón, J.M.; Ran, X.; Bouwman, A.F. Exploring spatiotemporal changes of the Yangtze River (Changjiang) nitrogen and phosphorus sources, retention and export to the East China Sea and Yellow Sea. Water Res. 2018, 142, 246–255. [Google Scholar] [CrossRef]
- Shen, D.; Huang, S.; Zhang, Y.; Zhou, Y. The source apportionment of N and P pollution in the surface waters of lowland urban area based on EEM-PARAFAC and PCA-APCS-MLR. Environ. Res. 2021, 197, 111022. [Google Scholar] [CrossRef] [PubMed]
- Pennino, M.J.; Kaushal, S.S.; Murthy, S.N.; Blomquist, J.D.; Cornwell, J.C.; Harris, L.A. Sources and transformations of anthropogenic nitrogen along an urban river–estuarine continuum. Biogeosciences 2016, 13, 6211–6228. [Google Scholar] [CrossRef]
- Chen, X.; Jiang, L.; Huang, X.; Cai, Z. Identifying nitrogen source and transport characteristics of the urban estuaries and gate-controlled rivers in northern Taihu Lake, China. Ecol. Indic. 2021, 130, 108035. [Google Scholar] [CrossRef]
- Bell, C.D.; Tague, C.L.; McMillan, S.K. Modeling runoff and nitrogen loads from a watershed at different levels of impervious surface coverage and connectivity to storm water control measures. Water Resour. Res. 2019, 55, 2690–2707. [Google Scholar] [CrossRef]
- Li, J.; Yu, S.; Qin, S. Removal capacities and environmental constrains of denitrification and Anammox processes in eutrophic riverine sediments. Water Air Soil Pollut. 2020, 231, 274. [Google Scholar] [CrossRef]
- Preisner, M. Surface water pollution by untreated municipal wastewater discharge due to a sewer failure. Environ. Process. 2020, 7, 767–780. [Google Scholar] [CrossRef]
- Zhang, M.; Chadwick, A.M. Nutrient dynamics at the sediment-water interface: Influence of wastewater effluents. Environ. Process. 2021, 8, 1337–1357. [Google Scholar] [CrossRef]
- Meyer, J.L.; Paul, M.J.; Taulbee, W.K. Stream ecosystem function in urbanizing landscapes. J. N. Am. Benthol. Soc. 2005, 24, 602–612. [Google Scholar] [CrossRef]
- Walsh, C.J.; Roy, A.H.; Feminella, J.W.; Cottingham, P.D.; Groffman, P.M.; Morgan, R.P. The urban stream syndrome: Current knowledge and the search for a cure. J. N. Am. Benthol. Soc. 2005, 24, 706–723. [Google Scholar] [CrossRef]
- Chadwick, M.A.; Dobberfuhl, D.R.; Benke, A.C.; Huryn, A.D.; Suberkropp, K.; Thiele, J.E. Urbanization affects stream ecosystem function by altering hydrology, chemistry, and biotic richness. Ecol. Appl. 2006, 16, 1796–1807. [Google Scholar] [CrossRef]
- Pereda, O.; Solagaistua, L.; Atristain, M.; de Guzmán, I.; Larrañaga, A.; von Schiller, D.; Elosegi, A. Impact of wastewater effluent pollution on stream functioning: A whole-ecosystem manipulation experiment. Environ. Pollut. 2020, 258, 113719. [Google Scholar] [CrossRef]
- Gu, C.; Waldron, S.; Bass, A.M. Carbon dioxide, methane, and dissolved carbon dynamics in an urbanized river system. Hydrol. Process. 2021, 35, 14360. [Google Scholar] [CrossRef]
- Assaad, A.; Pontvianne, S.; Pons, M.N. Assessment of organic pollution of an industrial river by synchronous fluorescence and UV–vis spectroscopy: The Fensch River (NE France). Environ. Monit. Assess. 2017, 189, 229. [Google Scholar] [CrossRef] [PubMed]
- Mineau, M.M.; Wollheim, W.M.; Buffam, I.; Findlay, S.E.G.; Hall, R.O.; Hotchkiss, E.R.; Koenig, L.E.; McDowell, W.H.; Parr, T.B. Dissolved organic carbon uptake in streams: A review and assessment of reach-scale measurements. J. Geophys. Res. Biogeosci. 2016, 121, 2019–2029. [Google Scholar] [CrossRef]
- Manning, D.P.; Rosemond, A.D.; Gulis, V.; Benstead, J.P.; Kominoski, J.S. Nutrients and temperature additively increase stream microbial respiration. Glob. Change Biol. 2018, 24, 233–247. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.A.; Ganf, G.G.; Brookes, J.D. A comparison of phosphorus and DOC leachates from different types of leaf litter in an urban environment. Freshw. Biol. 2008, 53, 1902–1913. [Google Scholar] [CrossRef]
- Worrall, F.; Howden, N.J.; Burt, T.P.; Bartlett, R. Declines in the dissolved organic carbon (DOC) concentration and flux from the UK. J. Hydrol. 2018, 556, 775–789. [Google Scholar] [CrossRef]
- Wen, Z.; Song, K.; Liu, G.; Lyu, L.; Shang, Y.; Fang, C.; Du, J. Characterizing DOC sources in China’s Haihe River basin using spectroscopy and stable carbon isotopes. Environ. Pollut. 2020, 258, 113684. [Google Scholar] [CrossRef]
- Lavelle, A.M.; Bury, N.R.; O’Shea, F.T.; Chadwick, M.A. Influence of urban river restoration on nitrogen dynamics at the sediment-water interface. PLoS ONE 2019, 14, e0212690. [Google Scholar] [CrossRef]
- Smith, L.; Watzin, M.C.; Druschel, G. Relating sediment phosphorus mobility to seasonal and diel redox fluctuations at the sediment–water interface in a eutrophic freshwater lake. Limnol. Oceanogr. 2011, 56, 2251–2264. [Google Scholar] [CrossRef]
- James, W.F. Nitrogen retention in a floodplain backwater of the upper Mississippi River (USA). Aquat. Sci. 2010, 72, 61–69. [Google Scholar] [CrossRef]
- Lin, X.; Li, X.; Gao, D.; Liu, M.; Cheng, L. Ammonium production and removal in the sediments of Shanghai river networks: Spatiotemporal variations, controlling factors, and environmental implications. J. Geophys. Res. Biogeosci. 2017, 122, 2461–2478. [Google Scholar] [CrossRef]
- Simon, K.S.; Chadwick, M.A.; Huryn, A.D.; Valett, H.M. Stream ecosystem response to chronic deposition of N and acid at the Bear Brook Watershed, Maine. Environ. Monit. Assess. 2010, 171, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Lu, S.Y.; Liao, Z.Y.; Ai, S.C.; Huang, M.S. Effect of mechanical aeration on nitrogen and microbial activity in sediment-water interface from urban lake. In Proceedings of the 2012 Asian Pacific Conference on Energy, Environment and Sustainable Development (APEESD 2012), Kuala Lumpur, Malaysia, 12–13 November 2012; pp. 770–775. [Google Scholar]
- Mulholland, P.J.; Helton, A.M.; Poole, G.; Hall, R.O.; Hamilton, S.K.; Peterson, B.J.; Tank, J.L.; Ashkenas, L.R.; Cooper, L.; Dahm, C.N.; et al. Stream denitrification across biomes and its response to anthropogenic nitrate loading. Nature 2008, 452, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Meghdadi, A. Characterizing the capacity of hyporheic sediments to attenuate groundwater nitrate loads by adsorption. Water Res. 2018, 140, 364–376. [Google Scholar] [CrossRef]
- Song, K.; Winters, C.; Xenopoulos, M.A.; Marsalek, J.; Frost, P.C. Phosphorus cycling in urban aquatic ecosystems: Connecting biological processes and water chemistry to sediment P fractions in urban stormwater management ponds. Biogeochemistry 2017, 132, 203–212. [Google Scholar] [CrossRef]
- Bedore, P.D.; David, M.B.; Stucki, J.W. Mechanisms of phosphorus control in urban streams receiving sewage effluent. Water Air Soil Pollut. 2008, 191, 217–229. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, Y.; Dou, X.; Yuan, H.; Yang, M. Granular ferric hydroxide adsorbent for phosphate removal: Demonstration preparation and field study. Water Sci. Technol. 2015, 72, 2179–2186. [Google Scholar] [CrossRef]
- Wu, P.; Yin, A.; Fan, M.; Wu, J.; Yang, X.; Zhang, H.; Gao, C. Phosphorus dynamics influenced by anthropogenic calcium in an urban stream flowing along an increasing urbanization gradient. Landsc. Urban Plan. 2018, 177, 1–9. [Google Scholar] [CrossRef]
- Martí, V.; Jubany, I.; Ribas, D.; Benito, J.A.; Ferrer, B. Improvement of phosphate adsorption kinetics onto ferric hydroxide by size reduction. Water 2021, 13, 1558. [Google Scholar] [CrossRef]
- Reitzel, K.; Hansen, J.; Andersen, F.Ø.; Hansen, K.S.; Jensen, H.S. Lake restoration by dosing aluminum relative to mobile phosphorus in the sediment. Environ. Sci. Technol. 2005, 39, 4134–4140. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.M.; Zhang, W.; Wang, X.X.; Zhou, Y.Y.; Yang, H.; Ji, G.L. Phosphorus in interstitial water induced by redox potential in sediment of Dianchi Lake, China. Pedosphere 2007, 17, 739–746. [Google Scholar] [CrossRef]
- Li, D.P.; Huang, Y.; Fan, X.C.; Yuan, Y. Contributions of phosphorus on sedimentary phosphorus bioavailability under sediment resuspension conditions. Chem. Eng. J. 2011, 168, 1049–1054. [Google Scholar]
- Zhang, Z.; Wang, Z.; Wang, Y.; Chen, X.; Wang, H.; Xu, X.; Lin, X.Y.; Czapar, G.F. Properties of phosphorus retention in sediments under different hydrological regimes: A laboratory-scale simulation study. J. Hydrol. 2011, 404, 109–116. [Google Scholar] [CrossRef]
- Li, X.; Zhou, Y.; Liu, G.; Lei, H.; Zhu, D. Mechanisms of the photochemical release of phosphate from resuspended sediments under solar irradiation. Sci. Total Environ. 2017, 595, 779–786. [Google Scholar] [CrossRef]
- Wang, J.; Xu, J.; Xia, J.; Wu, F.; Zhang, Y. A kinetic study of concurrent arsenic adsorption and phosphorus release during sediment resuspension. Chem. Geol. 2018, 495, 67–75. [Google Scholar] [CrossRef]
- Li, X.; Guo, M.; Duan, X.; Zhao, J.; Hua, Y.; Zhou, Y.; Liu, G.; Dionysiou, D.D. Distribution of organic phosphorus species in sediment profiles of shallow lakes and its effect on photo-release of phosphate during sediment resuspension. Environ. Int. 2019, 130, 104916. [Google Scholar] [CrossRef]
- Guo, M.; Li, X.; Song, C.; Liu, G.; Zhou, Y. Photo-induced phosphate release during sediment resuspension in shallow lakes: A potential positive feedback mechanism of eutrophication. Environ. Pollut. 2020, 258, 113679. [Google Scholar] [CrossRef]
- Wu, M.; Yang, F.; Yao, Q.; Bouwman, L.; Wang, P. Storm-induced sediment resuspension in the Changjiang River Estuary leads to alleviation of phosphorus limitation. Mar. Pollut. Bull. 2020, 160, 111628. [Google Scholar] [CrossRef]
- Wang, S.; Yi, W.; Yang, S.; Jin, X.; Wang, G.; Wu, F. Effects of light fraction organic matter removal on phosphate adsorption by lake sediments. Appl. Geochem. 2011, 26, 286–292. [Google Scholar] [CrossRef]
- Xie, F.; Dai, Z.; Zhu, Y.; Li, G.; Li, H.; He, Z.; Geng, S.; Wu, F. Adsorption of phosphate by sediments in a eutrophic lake: Isotherms, kinetics, thermodynamics and the influence of dissolved organic matter. Colloids Surf. A Physicochem. Eng. Asp. 2019, 562, 16–25. [Google Scholar] [CrossRef]
- Rodríguez-Castillo, T.; Estévez, E.; González-Ferreras, A.M.; Barquín, J. Estimating ecosystem metabolism to entire river networks. Ecosystems 2019, 22, 892–911. [Google Scholar] [CrossRef]
- Jankowski, K.J.; Mejia, F.H.; Blaszczak, J.R.; Holtgrieve, G.W. Aquatic ecosystem metabolism as a tool in environmental management. Wiley Interdiscip. Rev. Water 2021, 8, e1521. [Google Scholar] [CrossRef]
- Bernhardt, E.S.; Heffernan, J.B.; Grimm, N.B.; Stanley, E.H.; Harvey, J.W.; Arroita, M.; Appling, A.P.; Cohen, M.J.; McDowell, W.H.; Hall, R.O.; et al. The metabolic regimes of flowing waters. Limnol. Oceanogr. 2018, 63 (Suppl. 1), S99–S118. [Google Scholar] [CrossRef]
- Grace, M.; Imberger, S. Stream metabolism: Performing & interpreting measurements. In Murray Darling Basin Commission and New South Wales Department of Environment and Climate Change; Water Studies Centre Monash University: Melbourne, Australia, 2006; Volume 204, pp. 1–204. [Google Scholar]
- Tank, J.L.; Rosi-Marshall, E.J.; Griffiths, N.A.; Entrekin, S.A.; Stephen, M.L. A review of allochthonous organic matter dynamics and metabolism in streams. J. N. Am. Benthol. Soc. 2010, 29, 118–146. [Google Scholar] [CrossRef]
- Escoffier, N.; Bensoussan, N.; Vilmin, L.; Flipo, N.; Rocher, V.; David, A.; Métivier, F.; Groleau, A. Estimating ecosystem metabolism from continuous multi-sensor measurements in the Seine River. Environ. Sci. Pollut. Res. Int. 2018, 25, 23451–23467. [Google Scholar] [CrossRef]
- Roberts, B.J.; Mulholland, P.J.; Hill, W.R. Multiple scales of temporal variability in ecosystem metabolism rates: Results from 2 years of continuous monitoring in a forested headwater stream. Ecosystems 2007, 10, 588–606. [Google Scholar] [CrossRef]
- Heffernan, J.B.; Cohen, M.J. Direct and indirect coupling of primary production and diel nitrate dynamics in a subtropical spring-fed river. Limnol. Oceanogr. 2010, 55, 677–688. [Google Scholar] [CrossRef]
- Lupon, A.; Martí, E.; Sabater, F.; Bernal, S. Green light: Gross primary production influences seasonal stream N export by controlling fine-scale N dynamics. Ecology 2016, 97, 133–144. [Google Scholar] [CrossRef]
- Demars, B.O.; Manson, J.R.; Ólafsson, J.S.; Gíslason, G.M.; Friberg, N. Stream hydraulics and temperature determine the metabolism of geothermal icelandic streams. Knowl. Manag. Aquat. Ecosyst. 2011, 402, 5. [Google Scholar] [CrossRef]
- Beaulieu, J.; Arango, C.; Balz, D.; Shuster, W. Continuous monitoring reveals multiple controls on ecosystem metabolism in a suburban stream. Freshw. Biol. 2013, 58, 918–937. [Google Scholar] [CrossRef]
- Jauhiainen, J.; Kerojoki, O.; Silvennoinen, H.; Limin, S.; Vasander, H. Heterotrophic respiration in drained tropical peat is greatly affected by temperature-a passive ecosystem cooling experiment. Environ. Res. Lett. 2014, 9, 105013. [Google Scholar] [CrossRef]
- Williams, C.J.; Yamashita, Y.; Wilson, H.F.; Jaffé, R.; Xenopoulos, M.A. Unraveling the role of land use and microbial activity in shaping dissolved organic matter characteristics in stream ecosystems. Limnol. Oceanogr. 2010, 55, 1159–1171. [Google Scholar] [CrossRef]
- Ferreira, V.; Elosegi, A.; Tiegs, S.D.; Schiller, D.V.; Young, R. Organic matter decomposition and ecosystem metabolism as tools to assess the functional integrity of streams and rivers—A systematic review. Water 2020, 12, 3523. [Google Scholar] [CrossRef]
- Bernot, M.J.; Sobota, D.J.; Hall, R.O.; Mulholland, P.J.; Ddodds, W.K.; Webster, J.R.; Tank, J.L.; Ashkenas, L.R.; Cooper, L.W.; Dahm, C.N.; et al. Inter-regional comparison of land-use effects on stream metabolism. Freshw. Biol. 2010, 55, 1874–1890. [Google Scholar] [CrossRef]
- Dodds, W.K. Trophic state, eutrophication and nutrient criteria in streams. Trends Ecol. Evol. 2007, 22, 669–676. [Google Scholar] [CrossRef]
- Welti, N.; Striebel, M.; Ulseth, A.; Cross, W.F.; DeVilbiss, S.; Glibert, P.M.; Guo, L.; Hirst, A.G.; Hood, J.; Kominoski, J.S.; et al. Bridging food webs, ecosystem metabolism, and biogeochemistry using ecological stoichiometry theory. Front. Microbiol. 2017, 8, 1298. [Google Scholar] [CrossRef]
- Saunders, W.C.; Bouwes, N.; McHugh, P.; Jordan, C.E. A network model for primary production highlights linkages between salmonid populations and autochthonous resources. Ecosphere 2018, 9, e02131. [Google Scholar] [CrossRef]
- Yang, X.; Huang, S.; Wu, Q.; Zhang, R.; Liu, G. Diversity and vertical distributions of sediment bacteria in an urban river contaminated by nutrients and heavy metals. Front. Environ. Sci. Eng. 2013, 7, 851–859. [Google Scholar] [CrossRef]
- Belmeziti, A.; Cherqui, F.; Tourne, A.; Granger, D.; Werey, C.; Le Gauffre, P.; Chocat, B. Transitioning to sustainable urban water management systems: How to define expected service functions? Civ. Eng. Environ. Syst. 2015, 32, 316–334. [Google Scholar] [CrossRef]
- Wijesiri, B.; Liu, A.; Deilami, K.; He, B.; Hong, N.; Yang, B.; Zhao, X.; Ayoko, G.; Goonetilleke, A. Nutrients and metals interactions between water and sediment phases: An urban river case study. Environ. Pollut. 2019, 251, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Aristi, I.; von Schiller, D.; Arroita, M.; Barceló, D.; Ponsatí, L.; García-Galán, M.; Sabater, S.; Elosegi, A.; Acuña, V. Mixed effects of effluents from a wastewater treatment plant on river ecosystem metabolism: Subsidy or stress? Freshw. Biol. 2015, 60, 1398–1410. [Google Scholar] [CrossRef]
- Hou, P.; Ren, Y.; Zhang, Q.; Ye, Z.; Wang, H.; Fei, L. Temporal and spatial variations of water quality in a trans-urban river in Beijing, China. Fresenius Environ. Bull. 2013, 22, 561–572. [Google Scholar]
- Chen, X.; Zhou, W.; Pickett, S.T.A.; Li, W.; Han, L. Spatial-temporal variations of water quality and its relationship to land use and land cover in Beijing, China. Int. J. Environ. Res. Public Health 2016, 13, 449. [Google Scholar] [CrossRef]
- Liao, P.A. Analysis on water pollution trends and treatment technologies of the Beiyunhe Basin Beijing reach. Water Environ. 2013, 1, 16–19. (In Chinese) [Google Scholar]
- Pan, T.; Qi, J.; Wu, Q.; Wang, G.; Zhang, Y.P. Release law of nitrogen and phosphorus pollutants in sediments of Beiyun River Basin. Environ. Monit. China 2019, 35, 51–58. (In Chinese) [Google Scholar]
- Tian, G.S.; Chen, J. Study on water quality improvement measures and technology of the River Liangshui. Hai River Water Hydraul. 2008, 8, 46–49. (In Chinese) [Google Scholar]
- Bai, W.R. Analysis of water environment problems in the lower section of the River Tonghui. Water Environ. 2010, 1, 41–42. (In Chinese) [Google Scholar]
- Zhou, Z.X.; Xu, C.G.; Li, W.Y.; Yang, L.Q.; Sun, D.; Liu, J.H.; Liu, C. Current situation and problem analysis of water pollution in Changping section of the River Wenyu Basin. Water Environ. 2015, 4, 9–12. (In Chinese) [Google Scholar]
- Zhao, Z.; Liu, Q.; Leng, C.H. Integrated monitoring of heaven and earth of surface water environment in the Bahe River Basin in Chaoyang District of Beijing. China Soil Water Conserv. 2020, 6, 62–64. (In Chinese) [Google Scholar]
- Horie, Y.; Yonekura, K.; Suzuki, A.; Takahashi, C. Zinc chloride influences embryonic development, growth, and Gh/Igf-1 gene expression during the early life stage in zebrafish (Danio rerio). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2020, 230, 108684. [Google Scholar] [CrossRef] [PubMed]
- Heiri, O.; Lotter, A.; Lemcke, G. Loss on ignition as a method for estimating organic and carbonate content in sediments: Reproducibility and comparability of results. J. Paleolimnol. 2001, 25, 101–110. [Google Scholar] [CrossRef]
- Lee, J.Y.; Santamarina, J.C.; Ruppel, C. Parametric study of the physical properties of hydrate-bearing sand, silt, and clay sediments: 1. Electromagnetic properties. J. Geophys. Res. Solid Earth 2010, 115, 11. [Google Scholar] [CrossRef]
- Demars, B.L.; Thompson, J.; Manson, J.R. Stream metabolism and the open diel oxygen method: Principles, practice, and perspectives. Limnol. Oceanogr. Methods 2015, 13, 356–374. [Google Scholar] [CrossRef]
- Hill, N.B.; Riha, S.J.; Walter, M.T. Temperature dependence of daily respiration and reaeration rates during baseflow conditions in a northeastern U.S. stream. J. Hydrol. Reg. Stud. 2018, 19, 250–264. [Google Scholar] [CrossRef]
- Gillooly, J.F.; Brown, J.H.; West, G.B.; Savage, V.M.; Charnov, E.L. Effects of size and temperature on metabolic rate. Science 2001, 293, 2248–2251. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Chen, J.; Shirkey, G.; John, R.; Wu, S.R.; Park, H.; Shao, C. Applications of structural equation modelling (SEM) in ecological studies: An updated review. Ecol. Process. 2016, 5, 19. [Google Scholar] [CrossRef]
- Helton, A.M.; Poole, G.C.; Meyer, J.L.; Wollheim, W.M.; Peterson, B.J.; Mulholland, P.J.; Bernhardt, E.S.; Stanford, J.A.; Arango, C.; Ashkenas, L.R.; et al. Thinking outside the channel: Modeling nitrogen cycling in networked river ecosystems. Front. Ecol. Environ. 2011, 9, 229–238. [Google Scholar] [CrossRef] [Green Version]
- Reisinger, A.J.; Groffman, P.M.; Rosi-Marshall, E.J. Nitrogen-cycling process rates across urban ecosystems. FEMS Microbiol. Ecol. 2016, 92, 198. [Google Scholar] [CrossRef]
- Souza, A.C.; Pease, T.K.; Gardner, W.S. The direct role of enzyme hydrolysis on ammonium regeneration rates in estuarine sediments. Aquat. Microb. Ecol. Int. J. 2011, 65, 159–168. [Google Scholar] [CrossRef]
- Perryman, S.E.; Rees, G.N.; Walsh, C.J.; Grace, M.R. Urban stormwater runoff drives denitrifying community composition through changes in sediment texture and carbon content. Microb. Ecol. 2011, 61, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Monfared, S.A.H.; Darmian, M.D.; Snyder, S.A.; Azizyan, G.; Pirzadeh, B.; Moghaddam, M.A. Water quality planning in rivers: Assimilative capacity and dilution flow. Bull. Environ. Contam. Toxicol. 2017, 99, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Martí, E.; Riera, J.L.; Sabater, F. Effects of Wastewater Treatment Plants on Stream Nutrient Dynamics under Water Scarcity Conditions; Springer: Berlin/Heidelberg, Germany, 2009; pp. 173–195. [Google Scholar]
- Ratmaya, W.; Soudant, D.; Salmon-Monviola, J.; Plus, M.; Cochennec-Laureau, N.; Goubert, E.; Andrieux-Loyer, F.; Barillé, L.; Souchu, P. Reduced phosphorus loads from the Loire and Vilaine rivers were accompanied by increasing eutrophication in the Vilaine Bay (South Brittany, France). Biogeosciences 2019, 16, 1361–1380. [Google Scholar] [CrossRef]
- Pfeifer, L.R.; Bennett, E.M. Environmental and social predictors of phosphorus in urban streams on the Island of Montréal, Québec. Urban Ecosyst. 2011, 14, 485–499. [Google Scholar] [CrossRef]
- Jin, X.; Jiang, X.; Wang, Q.; Liu, D. Seasonal changes of P adsorption/desorption characteristics at the water sediment interface in Meiliang Bay, Taihu Lake, China. Acta Sci. Circumst. 2008, 28, 24–30. [Google Scholar]
- Luo, L.C.; Qin, B.Q.; Zhu, G.W. Calculation of total and resuspendable sediment volume in Lake Taihu. Oceanol. Limnol. Sin. 2004, 35, 491–496. [Google Scholar]
- Hale, R.L.; Turnbull, L.; Earl, S.R.; Childers, D.L.; Grimm, N.B. Stormwater infrastructure controls runoff and dissolved material export from arid urban watersheds. Ecosystems 2015, 18, 62–75. [Google Scholar] [CrossRef]
- Braissant, O.; Decho, A.W.; Dupraz, C.; Glunk, C.; Przekop, K.M.; Visscher, P.T. Exopolymeric substances of sulfate-reducing bacteria: Interactions with calcium at alkaline pH and implication for formation of carbonate minerals. Geobiology 2007, 5, 401–411. [Google Scholar] [CrossRef]
- Santomauro, G.; Baier, J.; Huang, W.; Pezold, S.; Bill, J. Formation of calcium carbonate polymorphs induced by living microalgae. J. Biomater. Nanobiotechnol. 2012, 3, 720–726. [Google Scholar] [CrossRef]
- Merbt, S.N.; Auguet, J.C.; Blesa, A.; Martí, E.; Casamayor, E.O. Wastewater treatment plant effluents change abundance and composition of ammonia-oxidizing microorganisms in Mediterranean urban stream biofilms. Microb. Ecol. 2015, 69, 66–74. [Google Scholar] [CrossRef]
- Bernal, S.; Segarra, A.; Merbt, S.N.; Martí, E. Differences in ammonium oxidizer abundance and N uptake capacity between epilithic and epipsammic biofilms in an urban stream. Freshw. Sci. 2017, 37, 13–22. [Google Scholar] [CrossRef]
- Jaiswal, D.; Pandey, U.; Mishra, V.; Pandey, J. Integrating resilience with functional ecosystem measures: A novel paradigm for management decisions under multiple-stressor interplay in freshwater ecosystems. Glob. Change Biol. 2021, 27, 3699–3717. [Google Scholar] [CrossRef] [PubMed]
- Pollock, M.S.; Clarke, L.M.J.; Dubé, M.G. The effects of hypoxia on fishes: From ecological relevance to physiological effects. Environ. Rev. 2007, 15, 1–14. [Google Scholar] [CrossRef]
- Rode, M.; Wade, A.J.; Cohen, M.J.; Hensley, R.; Bowes, M.; Kirchner, J.W.; Arhonditsis, G.; Jordan, P.; Kronvang, B.; Halliday, S.; et al. Sensors in the stream: The high-frequency wave of the present. Environ. Sci. Technol. 2016, 50, 10297–10307. [Google Scholar] [CrossRef] [PubMed]
- Elosegi, A.; Gessner, M.O.; Young, R.G. River doctors: Learning from medicine to improve ecosystem management. Sci. Total Environ. 2017, 595, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Young, R.G.; Matthaei, C.D.; Townsend, C.R. Organic matter breakdown and ecosystem metabolism: Functional indicators for assessing river ecosystem health. J. N. Am. Benthol. Soc. 2008, 27, 605–625. [Google Scholar] [CrossRef]
- Hall, R.O.; Beaulieu, J.J. Estimating autotrophic respiration in streams using daily metabolism data. Freshw. Sci. 2013, 32, 507–516. [Google Scholar] [CrossRef]
- Yan, J.; Liu, J.; You, X.; Shi, X.; Zhang, L. Simulating the gross primary production and ecosystem respiration of estuarine ecosystem in North China with AQUATOX. Ecol. Model. 2018, 373, 1–12. [Google Scholar] [CrossRef]
- Hung, J.J.; Wang, Y.J.; Tseng, C.M.; Chen, Y.L.L. Controlling mechanisms and cross linkages of ecosystem metabolism and atmospheric CO2 flux in the northern South China Sea. Deep. Sea Res. Part I Oceanogr. Res. Pap. 2020, 157, 103205. [Google Scholar] [CrossRef]
- Gücker, B.; Brauns, M.; Pusch, M.T. Effects of wastewater treatment plant discharge on ecosystem structure and function of lowland streams. J. N. Am. Benthol. Soc. 2006, 25, 313–329. [Google Scholar] [CrossRef]
- Griffiths, N.A.; Tank, J.L.; Royer, T.V.; Roley, S.S.; Rosi-Marshall, E.J.; Whiles, M.R.; Beaulieu, J.J.; Johnson, L.T. Agricultural land use alters the seasonality and magnitude of stream metabolism. Limnol. Oceanogr. 2013, 58, 1513–1529. [Google Scholar] [CrossRef]
- Hall, R.O.; Yackulic, C.B.; Kennedy, T.A.; Yard, M.D.; Rosi-Marshall, E.J.; Voichick, N.; Behn, K.E. Turbidity, light, temperature, and hydropeaking control primary productivity in the Colorado River, Grand Canyon. Limnol. Oceanogr. 2015, 60, 512–526. [Google Scholar] [CrossRef]
- Julian, J.P.; Doyle, M.W.; Powers, S.M.; Stanley, E.H.; Riggsbee, J.A. Optical water quality in rivers. Water Resour. Res. 2008, 44, 10411. [Google Scholar] [CrossRef]
- Izagirre, O.; Agirre, U.; Bermejo, M.; Pozo, J.; Elosegi, A. Environmental controls of whole-stream metabolism identified from continuous monitoring of Basque streams. J. N. Am. Benthol. Soc. 2008, 27, 252–268. [Google Scholar] [CrossRef]
- Demars, B.O.; Manson, J.R.; Ólafsson, J.S.; Gíslason, G.M.; Gudmundsdottir, R.; Woodward, G.; Reiss, J.; Pichler, D.E.; Rasmussen, J.J.; Friberg, N. Temperature and the metabolic balance of streams. Freshw. Biol. 2011, 56, 1106–1121. [Google Scholar] [CrossRef]
- Reid, A.J.; Carlson, A.K.; Creed, I.F.; Eliason, E.J.; Gell, P.A.; Johnson, P.J.; Kidd, K.A.; MacCormack, T.J.; Olden, J.D.; Ormerod, S.J.; et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol. Rev. Camb. Philos. Soc. 2019, 94, 849–873. [Google Scholar] [CrossRef]
- Perkins, D.M.; Yvon-Durocher, G.; Demars, B.O.; Reiss, J.; Pichler, D.E.; Friberg, N.; Trimmer, M.; Woodward, G. Consistent temperature dependence of respiration across ecosystems contrasting in thermal history. Glob. Change Biol. 2012, 18, 1300–1311. [Google Scholar] [CrossRef]
- Song, C.; Dodds, W.K.; Rüegg, J.; Argerich, A.; Baker, C.L.; Bowden, W.B.; Douglas, M.M.; Farrell, K.J.; Flinn, M.B.; Garcia, E.A.; et al. Continental-scale decrease in net primary productivity in streams due to climate warming. Nat. Geosci. 2018, 11, 415–420. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Delaney-Newcomb, K.; Findlay, S.E.G.; Newcomer, T.A.; Duan, S.; Pennino, M.J.; Sivirichi, G.M.; Sides-Raley, A.M.; Walbridge, M.R.; Belt, K.T. Longitudinal patterns in carbon and nitrogen fluxes and stream metabolism along an urban watershed continuum. Biogeochemistry 2014, 121, 23–44. [Google Scholar] [CrossRef]
- Levi, P.; McIntyre, P. Ecosystem responses to channel restoration decline with stream size in urban river networks. Ecol. Appl. 2020, 30, e02107. [Google Scholar] [CrossRef]









| Site | Season | DO (mg/L) | Reaeration (mgO2/(L·d)) | GPP (mgO2/(L·d)) | ER (mgO2/(L·d)) | NEP (mgO2/(L·d)) | Activation Energy (eV) |
|---|---|---|---|---|---|---|---|
| L1 | Autumn | 7.0 (4.3–10.0) | 5.3 (−5.3–14.2) | 13.3 (0–57.9) | 24.7 (21.3–27.9) | −11.4 (−27.9–32.8) | 0.57 (0.56–0.57) |
| Winter | 5.7 (3.9–7.9) | 6.2 (3.2–8.8) | 4.4 (0–19.2) | 8.8 (7.4–11.1) | −4.4 (–9.5–8.8) | 0.54 (0.54–0.55) | |
| Spring | 6.7 (5.3–8.7) | 13.1 (2.2–19.7) | 8.3 (0–28.1) | 22.3 (20.7–24.4) | −14.0 (–23.2–5.6) | 0.56 (0.56–0.56) | |
| Summer | 8.0 (4.6–11.4) | −1.3 (−6.5–3.9) | 20.6 (0–53.2) | 16.5 (14.4–19.0) | 4.1 (−16.9–35.1) | 0.58 (0.57–0.58) | |
| L2 | Autumn | 7.8 (4.9–11.1) | 1.5 (−3.2–5.9) | 12.0 (0–33.9) | 17.5 (16.3–18.7) | −5.5 (−18.2–15.6) | 0.57 (0.56–0.57) |
| Winter | 6.2 (5.0–7.6) | 6.8 (4.6–8.7) | 2.3 (0–12.0) | 6.9 (6.1–7.8) | −4.6 (−7.1–5.1) | 0.54 (0.54–0.55) | |
| Spring | 7.2 (5.5–9.0) | 2.2 (−0.7–4.7) | 8.3 (0–24.7) | 13.6 (12.8–14.5) | −5.3 (−14.5–11.0) | 0.57 (0.57–0.58) | |
| Summer | 8.9 (7.3–10.7) | −4.0 (−11.6–2.2) | 17.1 (0–46.0) | 13.7 (12.6–15.1) | 3.4 (−13.9–31.0) | 0.58 (0.57–0.58) | |
| T1 | Autumn | 7.0 (4.1–10.5) | 2.3 (−2.2–5.6) | 9.6 (0–29.6) | 15.5 (14.5–17.2) | −5.8 (−15.7–12.5) | 0.56 (0.56–0.57) |
| Winter | 6.9 (5.5–8.6) | 5.5 (2.5–7.7) | 2.8 (0–10.3) | 7.9 (7.3–8.9) | −5.1 (−8.3–1.4) | 0.55 (0.55–0.55) | |
| Spring | 8.5 (6.9–10.3) | 0.4 (−4.3–4.5) | 7.6 (0–25.8) | 11.2 (10.3–12.6) | −3.6 (–11.9–14.0) | 0.57 (0.56–0.57) | |
| Summer | 9.1 (7.3–11.3) | −3.3 (−10.4–2.0) | 13.4 (0–45.3) | 12.0 (10.3–14.6) | 1.4 (−13.5–31.7) | 0.58 (0.57–0.58) | |
| T2 | Autumn | 8.4 (6.8–10.3) | 1.5 (−4.1–5.6) | 8.4 (0–30.5) | 11.8 (11.0–13.0) | −3.4 (−12.2–17.6) | 0.56 (0.56–0.56) |
| Winter | 6.8 (5.6–8.4) | 4.6 (2.2–6.3) | 1.9 (0–9.9) | 6.9 (6.4–7.8) | −5.0 (−7.3–2.1) | 0.55 (0.55–0.55) | |
| Spring | 8.5 (6.7–10.5) | 0.02 (−1.3–1.3) | 8.5 (0–23.9) | 11.1 (10.7–12.1) | −2.6 (−11.1–12.3) | 0.57 (0.57–0.57) | |
| Summer | 10.3 (8.7–12.1) | −8.6 (−16.3–1.6) | 19.7 (0–51.0) | 8.6 (7.5–10.0) | 11.1 (−8.8–41.2) | 0.57 (0.57–0.58) | |
| W1 | Autumn | 7.8 (6.5–9.7) | 1.5 (−2.1–3.8) | 6.1 (0–25.3) | 8.2 (7.6–9.5) | −2.1 (−8.8–15.8) | 0.56 (0.56–0.57) |
| Winter | 6.1 (4.9–7.5) | 6.9 (4.6–8.7) | 1.9 (0–7.4) | 6.4 (5.8–7.5) | −4.5 (−7.0–0.8) | 0.55 (0.54–0.55) | |
| Spring | 8.0 (6.6–9.6) | 1.7 (−0.4–3.5) | 6.0 (0–16.2) | 8.0 (7.3–9.0) | −1.9 (−8.5–8.1) | 0.56 (0.55–0.56) | |
| Summer | 9.8 (8.2–11.4) | −3.3 (−7.1–0.3) | 15.1 (0–42.2) | 10.4 (8.7–12.6) | 4.7 (−10.7–31.1) | 0.57 (0.57–0.58) | |
| W2 | Autumn | 7.7 (6.2–9.8) | 2.0 (−2.6–5.1) | 5.7 (0–16.6) | 10.6 (9.5–12.9) | −4.9 (−11.4–5.5) | 0.56 (0.56–0.57) |
| Winter | 6.0 (4.8–7.7) | 7.8 (5.2–9.7) | 2.8 (0–9.8) | 7.2 (6.4–8.9) | –4.4 (−7.3–2.7) | 0.54 (0.54–0.55) | |
| Spring | 8.1 (6.9–9.4) | 2.2 (−0.2–4.3) | 5.2 (0–15.2) | 7.8 (7.1–8.6) | −2.6 (−8.4–6.6) | 0.56 (0.55–0.56) | |
| Summer | 9.3 (8.0–11.0) | 5.7 (−14.8–1.4) | 15.7 (0–50.7) | 7.7 (6.5–9.4) | 8.0 (−8.1–41.5) | 0.57 (0.57–0.58) |
| NO3− Concentration | NH4+ Concentration | PO43− Concentration | DOC Concentration | Water Temperature | DO | |
|---|---|---|---|---|---|---|
| NO3− flux F = 4.368 p = 0.042 | 𝛽 = −0.525 R2 = 0.292 P = 0.042 | |||||
| NH4+ flux F = 28.185 p < 0.001 | 𝛽 = 0.612 r2 = 0.612 p < 0.001 | |||||
| PO43− flux F = 6.194 P = 0.016 | 𝛽 = −0.622 r2 = 0.341 p = 0.016 | 𝛽 = −0.662 r2 = 0.453 p = 0.001 | ||||
| DOC flux F = 12.158 p = 0.001 | 𝛽 = −0.277 r2 = 0.113 p = 0.043 |
| Site | GPP (Min Mean–Max Mean) (gO2·m−2·d−1) | Impact | ER (Min Mean–Max Mean) (gO2·m−2·d−1) | Impact |
|---|---|---|---|---|
| L1 | 0.9–8.2 | No to severe effect | 1.8–7.4 | Mild to severe effect |
| L2 | 0.7–10.3 | No to severe effect | 2.1–8.8 | Mild to severe effect |
| T1 | 0.6–4.0 | Mild to severe effect | 1.6–4.6 | Severe effect |
| T2 | 0.8–13.8 | No to severe effect | 2.8–7.1 | Mild to severe effect |
| W1 | 0.6–9.1 | No to severe effect | 1.9–6.2 | Mild to severe effect |
| W2 | 1.1–12.5 | No to severe effect | 2.9–7.4 | Mild to severe effect |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, M.; Francis, R.A.; Chadwick, M.A. Nutrient Dynamics and Ecosystem Metabolism of Megacity Rivers: Influence of Elevated Nutrient Concentrations in Beijing’s Waterways. Water 2022, 14, 2963. https://doi.org/10.3390/w14192963
Zhang M, Francis RA, Chadwick MA. Nutrient Dynamics and Ecosystem Metabolism of Megacity Rivers: Influence of Elevated Nutrient Concentrations in Beijing’s Waterways. Water. 2022; 14(19):2963. https://doi.org/10.3390/w14192963
Chicago/Turabian StyleZhang, Meng, Robert A. Francis, and Michael A. Chadwick. 2022. "Nutrient Dynamics and Ecosystem Metabolism of Megacity Rivers: Influence of Elevated Nutrient Concentrations in Beijing’s Waterways" Water 14, no. 19: 2963. https://doi.org/10.3390/w14192963