Quality is Key
Emery Pharma’s Quality System ensures the quality, safety, and performance requirements for our Clients.
A dedicated Quality Assurance department implements all planned and systematic activities within the quality system. We produce high-quality, actionable data, to strengthen your regulatory package.
Stay in the Know
Emery Pharma scientists write about the work they have done and how they have helped companies like yours. Check out the full list of blogs here.
Native Mass Spectrometry (Native-MS) for Aggregate and Charge Variant Analysis
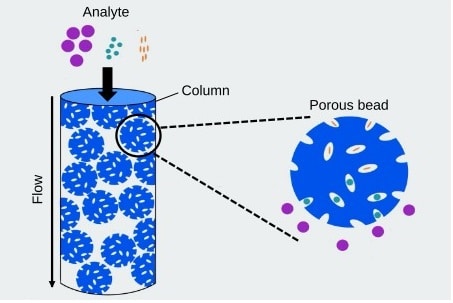
Size exclusion chromatography (SEC) and ion exchange chromatography (IEX) are traditional methods utilized for characterizing biotherapeutics under non-denaturing conditions. The typical high salt concentrations in their mobile phases make them…
A Guide to Quantitative NMR (qNMR)
Quantitative NMR (qNMR) stands as a robust method for gauging pharmaceutical purity. In the realm of analytical chemistry, nuclear magnetic resonance (NMR) spectroscopy emerges as a potent technique, offering insights…
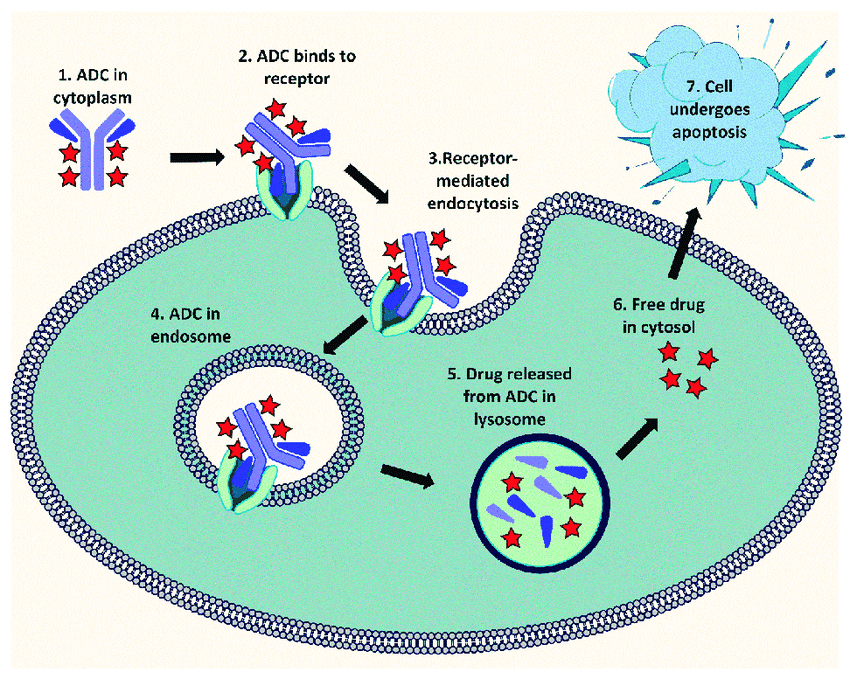
The Past, Present and Future of ADCs
ADCs, short for antibody-drug conjugates, consist of three essential components: a monoclonal antibody (mAb), a linker molecule, and a small molecule drug. In simple terms, an ADC is created by…